A few years back, during a family dinner, I was sitting opposite my uncle and I would not forget the moment that he was very carefully checking his blood sugar before he could eat. He had type 2 diabetes for more than ten years, and then he was undergoing a complex treatment consisting of several medicines, which never really worked out perfectly. Fast forward to today, and he’s on one injection a week, and his blood sugar control is better than it’s been in years.
It’s like my body remembered how to work again, he told me last month, his eyes genuinely bright with something I hadn’t seen before: hope.
That transformation? It’s largely thanks to our growing understanding of GLP and GIP in metabolism and how these hormones orchestrate one of the body’s most critical functions: insulin control. And honestly, the more I’ve learned about this, the more I realize we’re witnessing something pretty remarkable in medicine right now.
The Insulin Problem We’ve Been Trying to Solve
Let’s be real for a second. Insulin is kind of everything when it comes to metabolism. It’s the key that unlocks your cells so glucose can get in and provide energy. Without it, or when it doesn’t work properly, glucose just hangs out in your bloodstream, causing all sorts of trouble, nerve damage, vision problems, kidney issues, the whole nightmare scenario that comes with poorly controlled diabetes.
For decades, we’ve treated diabetes primarily by either giving people more insulin directly or trying to make their cells more sensitive to it. These strategies work to some extent. However, there was something that always bothered me about this strategy It felt as if we were just addressing the symptoms but not fully comprehending the process behind.
Then there are the GLP as well as GIP role in the control of blood sugar. These hormones incretin aren’t just part participants in the metabolic tale as they’re in fact the principal orchestra directors.
And the way they influence insulin? It’s smarter and more nuanced than we ever gave them credit for.
How GLP and GIP Control Insulin: The Beautiful Choreography
Okay, so here’s where it gets interesting: how GLP and GIP control insulin isn’t through some blunt force method. It’s elegant, really.
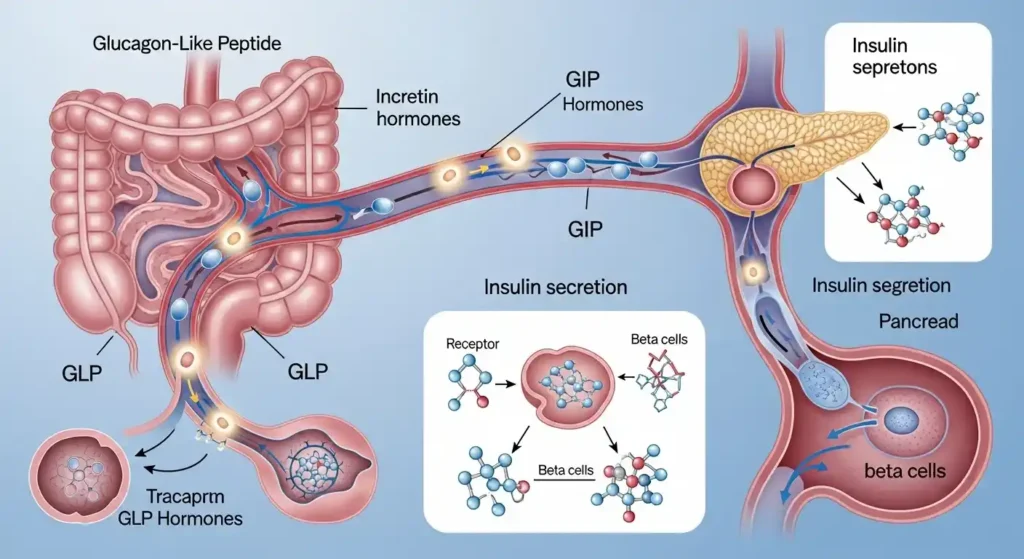
When you eat, especially foods with carbohydrates or fats, your gut releases these hormones. GIP shows up first from the upper intestine, then GLP-1 follows from further down the digestive tract. They travel through your bloodstream and find their way to your pancreas, where they bind to specific receptors on beta cells, those are the insulin factories.
But, and this is crucial, they don’t just flip a switch and say “make insulin.” The GLP GIP insulin response explained is that they amplify your body’s natural response to glucose. Think of it like this: if glucose is knocking on the door, GLP-1 and GIP are turning up the volume on that knock so the beta cells can hear it loud and clear.
What’s brilliant about this mechanism is that it’s glucose-dependent. The GLP GIP insulin secretion only really kicks into high gear when blood sugar is elevated. When your blood sugar is normal or low? These hormones back off. They’re not going to push your insulin levels up and cause dangerous hypoglycemia. Your body built in a safety feature.
Since the beginning of time until the present day, we’ve been evolving as a species to maximize nutritional intake. I am fascinated by how it evolved this way. It makes sense; during earlier stages of human evolution, there were periods of famine where food was not readily available; therefore, developing a system that could maximize nutrition when possible, while at the same time allowing for safe storage until the next available meal, allowed human beings to survive and thrive.
Blood Sugar Regulation: Real Life Impact
It’s been on my mind lately that when we think about glucose control in our bodies, we typically only think about it when something goes wrong. However, we constantly have to manage our glucose levels every moment of every day, whenever we eat. It’s an overwhelming thought when you think about how much work our pancreas does just to keep us alive! That’s not all, however, because the role of GLP and GIP in blood sugar regulation extends beyond just insulin secretion. GLP-1, in particular, has this multi-pronged approach that’s frankly kind of genius:
The first way that GLP–1 aids in managing glucose levels is by delaying gastric emptying. This means when we’re ready to eat, the stomach will actually take more time to empty its contents into the small intestine, which will cause glucose to be absorbed into the bloodstream at a slower, more even rate instead of causing a rapid increase in blood glucose. Ever notice how some meals give you steady energy while others lead to that awful sugar crash an hour later? Gastric emptying rate is part of that equation.
In addition, GLP-1 inhibits glucose, the hormone which tells your liver to let go of stored glucose. If you’ve eaten just now and you already have glucose flowing through your food items, you certainly shouldn’t require your liver to add additional sugar to the mixture. GLP-1 tells glucagon to sit this one out.
Third, and this one surprised me when I first learned about it, GLP-1 affects your brain. It crosses the blood-brain barrier and influences areas that control appetite and satiety. You feel fuller, satisfied sooner, and less inclined to reach for that second helping.
GIP works differently. It’s primarily focused on that insulin boost, but it also has some interesting effects on fat storage and possibly even bone metabolism. Scientists are still untangling all its roles, and I suspect we’ll discover even more in the coming years.
GLP and GIP in Diabetes Research: A Paradigm Shift
The story of GLP and GIP in diabetes research is one of those scientific journeys that sounds obvious in hindsight but took decades to fully appreciate.
Researchers noticed something weird back in the 1960s: when you gave someone glucose orally, they produced more insulin than when you gave them the same amount of glucose intravenously. Same glucose, same blood sugar spike, but different insulin responses. They called this the “incretin effect.”
It took years to identify GLP-1 and GIP as the main incretin hormones. And then came the heartbreaking discovery: people with type 2 diabetes have a severely diminished incretin effect. Their GLP-1 and GIP responses are blunted. It’s as if their gut and pancreas have ceased to communicate properly.
That realization opened up entirely new therapeutic avenues. Instead of just throwing more insulin at the problem, what if we could restore that natural gut-pancreas conversation?
The GLP and GIP medical importance suddenly became crystal clear. These weren’t just interesting biological curiosities; they were potential game-changers for millions of people struggling with metabolic disease.
The Rise of GLP GIP Therapy: Benefits, Realities, and What We’re Still Learning
So GLP GIP therapy has basically exploded in the past few years, and I mean exploded. You can’t scroll through social media without seeing someone talking about these medications.
The first wave came with GLP-1 receptor agonist drugs that mimic the action of natural GLP-1 but last much longer in the body. Drugs like exenatide, liraglutide, and semaglutide. The GLP GIP benefits were impressive right out of the gate: better blood sugar control, less hypoglycemia risk compared to insulin, and here’s the kicker that made headlines — significant weight loss.
My uncle’s on semaglutide, and his A1C (that’s the three-month average blood sugar marker) has dropped from 9.2 to 6.8. That’s moving from poorly controlled diabetes to near-normal levels. His weight’s down 35 pounds. His doctor is thrilled. He’s thrilled.
But then researchers thought: what if we combined GLP-1 effects with GIP effects? That led to dual agonists like tirzepatide, which stimulates both GLP-1 and GIP receptors. The results have been even more striking; some people are seeing A1C reductions of 2-2.5 percentage points and weight loss averaging 15-20% of body weight.
It’s hard not to get excited about those numbers. But I’d be lying if I said there weren’t considerations.
GLP GIP side effects are real and, for some people, pretty unpleasant. Nausea tops the list, sometimes severe nausea that makes eating difficult. There’s vomiting, diarrhea, and constipation. These usually improve over time as your body adjusts, but the initial weeks can be rough. I’ve heard stories from friends who’ve tried these medications and couldn’t tolerate them, others who pushed through and found the side effects manageable.
There are also questions we’re still answering. What are the long-term effects of being on these medications for years or decades? We have solid data for a few years now, but metabolic medicine is a marathon, not a sprint. What happens if you stop the medication? For many people, weight comes back, and blood sugar control deteriorates. Is that a problem with the drug or a statement about the chronic nature of metabolic disease?
And let’s talk about access. These medications are expensive, we’re talking over a thousand dollars a month without insurance. Insurance coverage is inconsistent. Some people who would genuinely benefit can’t access them. That’s not a scientific problem; that’s a healthcare system problem, but it’s part of the conversation we need to have about GLP GIP medical trend.
GLP and GIP medical importance
The GLP and GIP medical importance extends beyond diabetes and obesity, I think. Researchers are exploring potential benefits for cardiovascular disease, fatty liver disease, kidney protection, and even neurodegenerative conditions. The more we understand these hormones’ roles throughout the body, the more potential applications we’re finding.
There’s also research going in opposite directions simultaneously, which I find fascinating. Scientists are looking into GIP antagonists, which block GIP instead of activating it. They are also combining GLP-1 agonism. Some are doubling down and creating triple agonists which are able to target GLP-1 GIP as well as the glucagon receptors. It’s still an experiment stage with lots of this. We’re trying to figure out which combinations work the best with which types of individuals.
This personalization component is likely the next frontier. Not everyone responds the same way to these medications. You lose weight very quickly one week and not at all the next. Some people’s diabetes goes into remission, and for others, it just gets a little better. Understanding why those differences exist, genetics, gut microbiome, and prior metabolic history, that’s the next frontier.
A Personal Reflection
Honestly, I keep coming back to my uncle. No one wants to see someone they love struggle and watch them struggle with a disease for a long time, going through so many medications, and balancing the mental load with all the side effects and with the daily complications of just trying to manage it all, and then finally they find a solution that works. It’s astonishing.
The science of GLP and GIP in metabolism isn’t just academic for millions of people. It’s the difference between having your body control you versus having a sense of control over your body; between always worrying about what’s going to happen next versus feeling cautiously optimistic about what’s going to happen next. Are these medications a cure-all? No. Are they going to be beneficial to everyone? But they represent a fundamental shift in how we understand the conditions and treat them, and that matters.
We are moving away from symptom-focused treatments to working in concert with the body’s self-regulatory mechanisms. We’re learning to speak the language the gut and pancreas use to communicate. And just maybe, we’re getting closer to helping people not simply manage disease but genuinely feel better.
This is opening the door to understanding how the gut and pancreas communicate with one another, and perhaps through this understanding, we’ll be able to help people not only control their disease but actually feel better. Because at the end of the day, behind every statistic about A1C reduction or percentage weight loss, there’s a person. Someone’s parent, sibling, or friend. Someone who deserves to feel good in their body, to not be constantly exhausted by the management of chronic disease, and to hope for more good years ahead.
The insulin connection we’re unraveling through GLP and GIP? It’s not just changing metabolic medicine; it’s quietly changing lives for many people. And I think that’s a reason to celebrate, even as we continue asking hard questions and pushing for more.